Kevin Litchfield: using whole exome sequencing in testicular cancer
Seven Bridges Fridays
Each week our London office holds Seven Bridges Fridays, where we invite speakers from academia and industry to discuss their work over beer and snacks.
Our recent presenter was a mathematician turned bioinformatician who worked in the pharmaceutical industry for 8 years. Kevin Litchfield is now finishing his PhD in Molecular Biology at the Institute for Cancer Research in London. Kevin’s research focuses on the genomic architecture of testicular germ cell tumors (TGCTs).

Genetics of testicular cancer
Testicular cancer is the most common cancer in men aged 15–45 years, with over 95% of cases being TGCTs. Over 18,000 TGCTs are diagnosed each year in Europe, and the number of cases has doubled in the past 40 years. Studies of families and twins show a strong genetic basis for TGCTs with brothers of patients being at eight times increased risk to also develop TGCT. Extra copies of some chromosomes, typically 7, 8, 21, 22 and X are often present in these tumors.
Mutations associated with testicular cancer are not Y chromosome linked, making the disease inheritable from both parents. Kevin explained that similar mutations in females lead to ovarian disease.
Cancer cell exome sequencing
Analysis of whole exome sequencing data from 42 TGCTs enabled Kevin and his collaborators to identify additional genetic characteristics of this disease. The rate of mutation for the tumors was uniformly low, which is expected given their germ cell origin. Recurrent mutations in the tumor suppressor gene CDC27 and increased copy numbers of the spermatocyte development gene FSIP2 were uncovered. Mutations in XRCC2, a gene involved in resistance to the antitumor agent cisplatin, were identified in samples from the two patients who did not respond to chemotherapy.
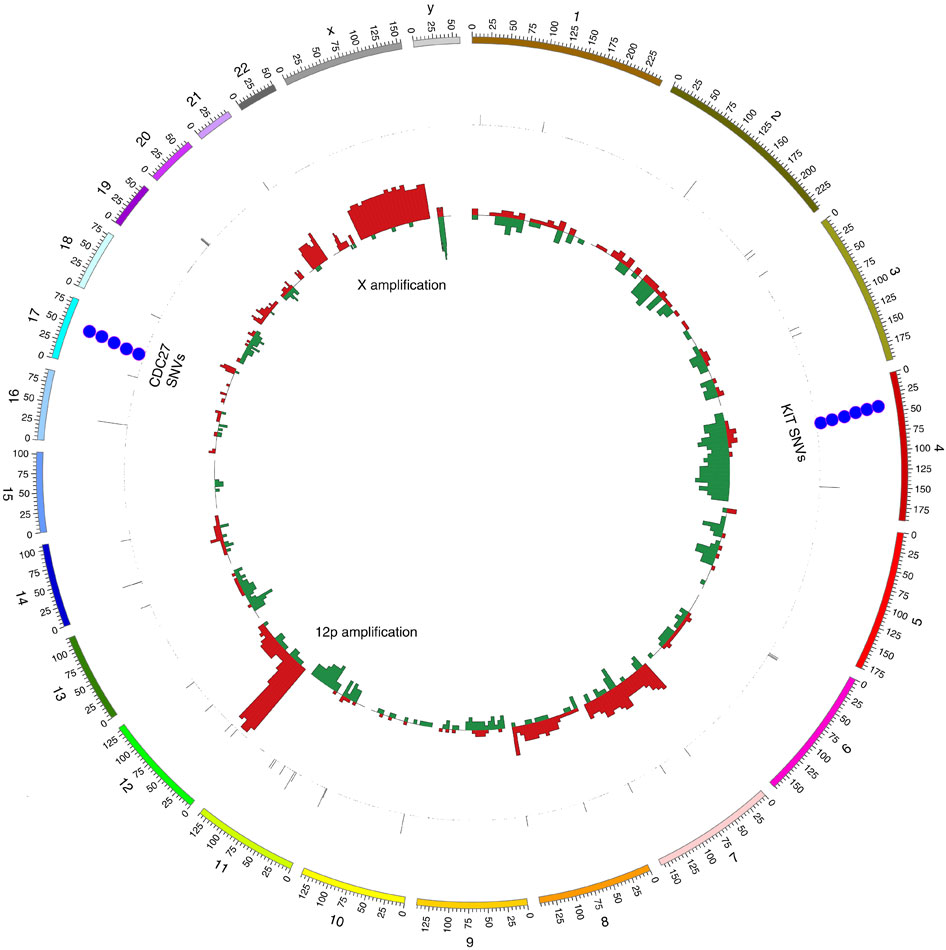
Kevin also talked about four new susceptibility loci for TGCT identified in a genome-wide association study in >25,000 men. These loci, together with previously known single nucleotide polymorphisms support a polygenic model for TGCT risk. Kevin explained the biological basis for the development of this disease is likely to involve the sex determination pathway, KIT/KITLG signalling, centrosome cycle, and DNA repair.
The future
Given the low incidence of disease, the effectiveness of current treatments, and the current cost of genetic screening, it is hard to make a case for population-level genetic screening for TGCT. Screening people with a known high risk of developing the disease might, however, be beneficial. Future advances such as routine generation of full genomic data for patients is likely to change this.
Big thanks to Kevin for visiting and showing his work!
Was great to meet the SBG team – keep the good work! https://t.co/tgh2MbEQLx
— Kevin Litchfield (@kevlitchfield) April 30, 2016
Kevin encouraged fundraising support for the Movember Foundation, which works to support men and boys living with and beyond testicular cancer.
Kevin’s presentation was part of the Seven Bridges Fridays series, held at our London offices near Kings Cross. Drop us a line if you’d like to stop by to meet the team, or if you’d like to present something about genomics, medicine, bioinformatics, cloud computing (or basically anything we’ll find interesting and can discuss over a beer)!
This post was written by Andrea Szöllössi, who came to Seven Bridges Fridays and ended up joining us as a Technical Writer.